An Alzheimer’s Warning 25 Years Before Symptoms Show
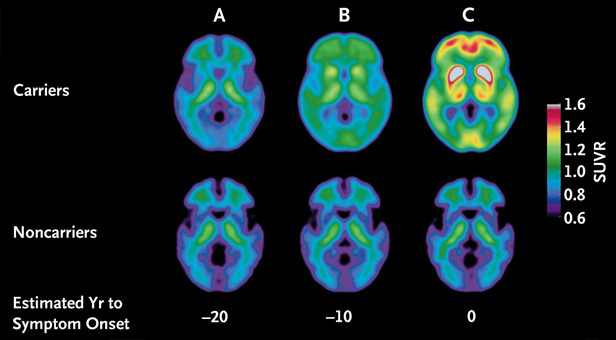
The first detectable signs of Alzheimer’s disease occur as long as a quarter century before symptoms like memory loss become noticeable, according to a detailed chronology of molecular changes to the brain and spinal fluid of people who later developed the brain disease.
The research, published today in the New England Journal of Medicine, provides a timeline of the subtle changes that begin in victims’ brains and, importantly, can be detected years ahead of time by MRI exams, blood analyses, or other tests.
The development of biomarkers that can track and predict the natural course of the disease is important for carrying out drug studies, in part because changes to these molecules could give early hints that a drug works. Treatments for Alzheimer’s have all been unsuccessful so far—in part, researchers think, because people received drugs only after symptoms had become obvious and their brains were too damaged to recover.
“This is perhaps the most comprehensive picture of what changes, in what order, and by how much,” says Randall Bateman, a professor of neurology at Washington University School of Medicine, and the study’s first author.
Now, with a roadmap of the disease’s signature, Bateman says, doctors will have a better chance of judging whether a treatment is working by looking at interim measures rather than waiting until the disease is full-blown. Bateman is participating in a set of important new studies to see if Alzheimer’s can be prevented. The two-year drug trials involve currently healthy patients who carry genes for an early-onset, inherited form of Alzheimer’s. The biomarkers will be used to judge whether the medications are helpful at preventing or slowing the course of the disease.
A combination of factors similar to those that lead to heart disease—diabetes, poor diet, lack of exercise—are associated with the development of Alzheimer’s, although the disease’s root cause is still a matter of scientific debate.
The timeline was constructed from studies of 128 people from families plagued by a rare, inherited form of Alzheimer’s in which symptoms emerge in patients still in their 30s, instead of after 65, as is the case with most of the five million Americans with the disease.
According to the study, levels in the spinal fluid of Aβ42, a protein associated with Alzheimer’s, begins to decline 25 years before the onset of symptoms; at 15 years before symptoms develop, levels of another protein, called tau, begin to rise, and some brain shrinkage and atrophy is evident; at 10 years out, the brain’s consumption of the sugar glucose is discernibly lower, and some memory impairment can be measured.
The researchers created the chronology by measuring the key molecules in volunteers who are healthy, but whose genes predict they will develop Alzheimer’s. By learning at what age those volunteers’ parents first developed Alzheimer’s symptoms, the researchers were able to infer the time span between the molecular changes and the disease’s onset.
Family members who did not inherit the Alzheimer’s genes showed none of these early signs of the disease, Bateman says, suggesting that the markers really do distinguish between people with early-stage disease and those without. It remains to be seen whether the biomarkers can also predict Alzheimer’s disease in the general population.
The Washington University scientists continue to look for more families with the genetic form of Alzheimer’s to join its study, which is recruiting families with a history of early-onset Alzheimer’s spanning three generations.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.