From the Labs: Biomedicine
Glowing Monkeys
Primates pass a fluorescence gene to their offspring.
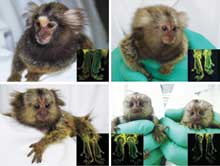
Source: “Generation of transgenic non-human primates with germline transmission”
Erika Sasaki, Hiroshi Suemizu, et al.
Nature 459: 523-527
Results: Scientists transferred a gene derived from jellyfish into marmoset monkeys, causing them to produce a protein that makes them glow green. The monkeys then passed the gene for the fluorescent protein to their offspring, which glow as well.
Why it matters: Genetically engineered mice have become common and vital tools for biomedical research. Now it’s possible, for the first time, to make genetically engineered strains of primates. Scientists could use the modified animals to study neurological diseases such as Parkinson’s, which cannot be adequately reproduced in rodents.
Methods: To create the transgenic monkeys, researchers injected a virus carrying the gene for green fluorescent protein, or GFP, into 91 marmoset embryos. Eighty healthy transgenic embryos were then transplanted into surrogate mothers, which gave birth to five glowing offspring. Three glowing second-generation marmosets have been born since April.
Next steps: The researchers are further refining their approach to deliver larger pieces of DNA and to block the action of specific genes. Both techniques will be necessary to develop marmoset models of human disease.
Heart Repair
Patients’ own bone marrow cells help relieve chest pain.
Source: “Intramyocardial bone marrow cell injection for chronic myocardial ischemia: a randomized controlled trial”
Jan van Ramshorst et al.
Journal of the American Medical Association 301: 1997-2004
Results: Three months after being injected with cells derived from their own bone marrow, patients with reduced blood supply to the heart muscle–a condition known as ischemic heart disease–had better blood flow to the heart, less chest pain, and better exercise capacity.
Why it matters: Previous studies testing the benefits of bone marrow cells on different types of heart disease have shown mixed results. This study is the first to show an improvement in blood flow, and it suggests that this type of cell therapy may be more effective in people with chronic heart conditions, such as angina, than in those who have suffered heart attacks.
Methods: Scientists extracted 80 milliliters of bone marrow from each patient and isolated mononuclear cells–a mix of stem cells and progenitor cells. Then they injected either a solution of these cells or a placebo into the heart muscle. After three months, the scientists used magnetic resonance imaging and other methods to assess blood flow to the heart. They also measured patients’ ability to exercise comfortably for a specified time and assessed their quality of life.
Next steps: The researchers are trying to determine which of the different cell types in bone marrow have the greatest benefit and how they improve blood flow–for example, by encouraging the growth of new blood vessels or improving the health of existing ones.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.